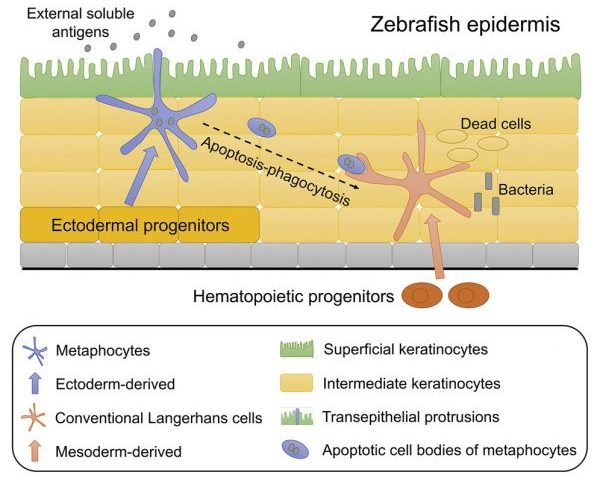
Tissue-resident macrophages (TRMs) are highly heterogeneous and engage in a wide range of diverse functions. Yet, the heterogeneities of their origins and distinct functions remain poorly defined.
Using a high temporal-spatial resolved cell labelling combined with transcriptome and functional analysis, Dr. Zilong Wen’s laboratory has recently identified a myeloid-like cell population in zebrafish epidermis. This newly identified myeloid-like cell population, designated as metaphocytes, are of ectodermal origin but shares high similarities with mesoderm-derived conventional macrophages in the skin – Langerhans cells in both anatomical location, cellular morphology and transcriptome profile.
Surprisingly, unlike conventional Langerhans cells, metaphocytes respond to neither injury nor bacteria challenge, but rather they capture soluble antigens from the external environment through transepithelial protrusions and transfer the antigens to Langerhans cells through apoptosis-phagocytosis cascade. These results indicate that metaphocytes may play a critical role in T cell activation in response to soluble antigens through regulating the antigen uptake of Langerhans cells.
This study, which recently published in Developmental Cell, documents the existence of ectoderm-derived myeloid-like cells and opens a new paradigm for investigation the heterogeneities of resident immune cells.
Journal Reference:
Xi Lin, Qiuxia Zhou, Changlong Zhao, Guanzhen Lin, Jin Xu, and Zilong Wen. An Ectoderm-Derived Myeloid-like Cell Population Functions as Antigen Transporters for Langerhans Cells in Zebrafish Epidermis. Dev Cell, 2019; DOI: 10.1016/j.devcel.2019.03.028